2.
POTENTIOMETRIC TITRATIONS
Electrodes used in potentiometric
titrations
The experimental setup for potentiometric measurement comprises a set of an indicating and reference electrodes or two identical indicating electrodes, which should be treated carefully. Do not place the electrodes anywhere except attached to the electrode holder. At the end of the experiment rinse the electrodes and place each one in its housing as required.
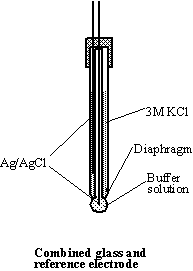
Glass electrode. Combined glass reference
electrode consists of indicator and reference electrodes in the same body.
Great care should be taken of it: never touch the glass part of the electrode
with anything except soft tissue paper. While in use, the bulb of the glass
electrode and the diaphragm of the reference electrode should be immersed in
solution. For short-term storage the combined glass electrode should be
immersed in solution of
Ion-selective electrodes are used for detection of specific ions in a mixture of ions. The sensor element, ion-selective membrane, has a construction similar to that of glass electrode. For calibration of ion-selective electrodes a standard addition method is often employed.

Silver
indicating electrodes are silver wires with 1-
Mercury-coated indicating electrodes are reported to be prepared by lightly amalgamating a gold wire. The disadvantage in use of gold is that it is consumed with time by the amalgam formation. Silver wire used instead of gold, however, can serve many years. The preparation of mercury-coated silver electrode is done by the instructor in a hood (mercury vapors are poisonous!). The silver wire (~1.5 mm diameter) is (a) rubbed with emery paper, rinsed with distilled water and dried with tissue; (b) dipped into mercury to form an amalgame; (c) the mercury is gently spread on the wire with soft tissue. This electrode may be used during several runs of titrations without any renewal. For renewal, step (a) may be omitted.
Platinum
redox electrodes are used in redox potentiometric titrations.
In excess of oxidant oxide films are formed on the platinum electrodes. The
potential response of the electrode is distorted, and the film must be removed.
Efficient pretreatment is achieved by cathodically
polarizing the platinum electrode in
Gold redox electrodes are seldom used in potentiometric titrations. According to our recent experience, the gold electrodes are better behaved than platinum electrodes in view of rate of response and stability toward formation of oxides. These features are of high importance in continuous mode of titration. A good example is the use of gold electrodes in the titration of ascorbic acid with bromine in continuous mode, where the response of the platinum electrode is unsatisfactory.
Reference
electrodes. Calomel and silver/silver-chloride electrodes are commonly
used in potentiometric titration. In the case of
possible interferences of chlorides (as in determination of halides), a mercurous sulfate electrode may be used. In the following
series of experiments a home made Ag/AgCl/1 M KCl reference electrode is used. Its potential is -19
mV vs SCE, at

Exp. 1. Compexometric
titration of Ca2+ and Mg2+ in drinking water
Chelating agents
A chelating agent (ligand) is a multidendate compexing molecule, forming usually a 1:1 complex (chelate) with a metal ion Mz+, regardless of the value of the charge z of the metal ion.
Complexometric titration using chelating agent Y (comprising n complexing groups) is far more advantageous than titration with monodendate ligand L (of the same type), for two reasons:
(1) The chelate MY is more
stable than the MLn complex due to
thermodynamic considerations. The free energy change, ![]() ,
,
![]()
is characterized by similar ![]() values, but very
different
values, but very
different ![]() values, were
values, were ![]() is the enthalpy and
is the enthalpy and ![]() is the entropy. More
disorder is created by the dissociation of MLn
with the formation of n+1 species, compared to two species for the chelate.
is the entropy. More
disorder is created by the dissociation of MLn
with the formation of n+1 species, compared to two species for the chelate.
(2) Stepwise complexation with monodendate ligands is usually characterized with successive formation constants relatively close to each other. Thus, there are several types of complexes in the vicinity of the end point, compared to a chelate, where a single 1:1 complex is usually formed.
One of the most widely used chelating agents is EDTA (ethylenediaminetetraacetic acid) with six complexing groups (two nitrogen and four carboxylic groups). EDTA titrations have been applied to the determination of most metal cations with the exception of the alkali metal ions. Selectivity is obtained by controlling the pH. Ca2+ and Mg2+ have however close formation constants (Kf = 5.0·1010 and 4.9·108 respectively), unsuitable for separate detection. Titrating with EDTA, the total concentration of Ca2+ and Mg2+ is determined.
Another chelating agent is EGTA, which is suitable for the determination of [Ca2+] alone, due to the large difference in Kf (1.0·1011 and 1.6·105 for Ca2+ and Mg2+, respectively).
The mercury electrode, Hg/HgY,
MY, Mn+
The mercury electrode can serve as an indicating electrode for complexometric titrations when it is used as an electrode of the third kind.
Let us consider a mercury electrode dipped in a solution containing the following relevant species: Hg2+, M2+ (metal ions), Y4-, HgY2-, MY2-.
The following processes are to be considered:

where K´f is the
conditional formation constant (cf., ref.
The expression for the equilibrium potential is obtained, combining the previous equations.
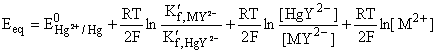
The potential of the mercury electrode varies with the variation of the concentrations of HgY2-, MY2- and M2+. Around the equivalence point, however, [MY2-] is almost constant (why?); [HgY2-] is virtually constant when the condition K'f,HgY2- >> K'f,MY2- is fulfilled (why?). Since HgY2- forms a very stable complex, the inequality is fulfilled for most metal ions. The concentration of M2+ varies by several orders of magnitude around the equivalence point.
Since the first three terms on the right-hand of the last equation are almost constant around the equivalence point, the electrode potential serves as reliable indicator for the variation of the concentration of the uncomplexed metal ion. Thus the electrode is an efficient tool for the end-point detection of complexometric titrations.
According to the above thermodynamic treatment, the presence of Hg(II) species is imperative for the proper functioning of the electrode. This is a reason that a small amount of the EDTA complex of Hg(II) is introduced into the solution in complexometric titrations (why Hg(II) in the complex form and not as Hg2+ is used?).
Chemicals 1.
2.
3.
4. Buffer pH 10.5 (
Indicating system: Mercury-coated silver electrode and reference electrode
Procedure
The titration is performed in the repetitive-monotonic mode (cf., Titrimetric Methods of Analysis, section 1.5).
Determination
of Ca2+ with EGTA: pipet 1.00 ml
of drinking water into the vessel. Add 1 ml of buffer pH 10.5, 1 drop
Determination of total Ca2+ and Mg2+: as above. Titrate with EDTA.
Calibrate the flow rate of the pump according to the instructions of section 1.6, Titrimetric Methods of Analysis, if needed.
Measure the distances between the successive end points in each series, calculate the average and the standard deviation. Compare the average with the result of the first titration.
Report the concentration of Ca2+ and Mg2+ in ppm and mol/l. Compare your results with the official permissible levels.
Exp. 2. Compexometric EDTA
titrations of metals
Chemicals
|
Analyte
metal |
|
|
|
|
|
|
|
+ |
+ |
|
+ |
|
|
|
+ |
+ |
+ |
|
|
|
|
+ |
+ |
|
|
+ |
|
|
+ |
+ |
|
|
+ |
Indicating system: Mercury-coated silver electrode and reference electrode
Procedure
Pipet to the vessel 0.500 ml of
Obtain an unknown sample and repeat the titration.
For each titration plot E vs V (potential vs volume).
Report the concentrations of the metal in the stock solution and in the unknown in mol/l.
Exp. 3. Automatic potentiometric
acid-base titration.
Determination of bicarbonates in
drinking water
The determination is based on a titration with HCl:
![]()
The concentration of bicarbonates in drinking water is in the level of millimolars.
For a monotonic automatic titration, the slow response of the glass electrode around the end point should be taken into consideration. If constant-rate titration is to be performed, the rate of the addition of the titrant should be very slow in order to allow the potential of the glass electrode to approach steady-state readings. The whole titration should be carried out in not less than 5 minutes.
Gran's treatment of
the titration curve
In the case of weak base - strong acid titration the treatment consists in linearization of the part beyond the equivalence point. The transformation of potential signal E to concentration of hydrogen ions [H+] is carried out by equation
![]()
The transformed function is (Vt/Vin)·10((E-k)/s), where Vt/Vin is a correction for dilution. For more details refer to Titrimetric Methods of Analysis, section 1.5.
For
the determination of the end point according to Gran's
treatment, one needs to know the initial volume of the solution in the
titrating vessel and the accurate values of k and s. The values of k and s are determined
from a blank titration, performed under the same conditions and at the time
close to the time of the titration of sample. For the blank titration a
solution with ionic strength similar to that of drinking water (5 -
Chemicals 1.
2.
3.
Electrode: Combined glass and reference electrode
Procedure
1. Calibrate, if needed, the flow rate of the pump according to the instructions in section 1.6, Titrimetric Methods of Analysis.
2. Pipet 5.00 ml of the bicarbonate sample into the cell (use the standard bicarbonate solution and/or the samples of drinking water provided). Start stirring. Add a measured volume of distilled water to just cover the electrode. Wait until the potential reading is stable. Dip the pump tube with the titrant into the solution. Operate the pump to start the titration. The titration curve is displayed on the recording device. At the end of the titration shut off the pump and the recording device.
3. Blank
titration - determination of k and s. Repeat the above titration
procedure using 5.00 ml
Treatment of results
1. Estimate the first dissociation constant of carbonic acid, Ka, on the basis of the titration curve of the bicarbonate solution.
2. Calculate the values of k and s.
3. Determine the end point: (a) from the first derivative of the titration curve and (b) according to Gran plot. Compare the two results.
4. Report the concentration of bicarbonate in ppm HCO3- and mol/l.
Exp. 4. Potentiometric
titration of Fe2+ with Ce4+.
End-point detection with dual
polarized electrodes
Chemicals
1.
2.
3.
4.
Electrodes: a) Platinum electrode and reference electrode
b) Twin platinum electrodes
Procedure
Pipet 5.00 ml of
Titrate
with standard
Plot the titration curves.
Report the concentration of the ferrous sulfate solution in mol/l.
Is it possible for the polarization current to change the composition of the solution?
Note:
1. For optimal
functioning of the platinum indicating electrodes, perform from time to time
the electrochemical pretreatment to remove the oxide film, which gradually forms
in presence of Ce4+. Follow the instructions in section
"Electrodes used in potentiometric
titrations".
2. For concentrations of Fe2+ lower than 10-
Exp. 5. Potentiometric determination of halides
Chemicals
1.
2.
3.
4.
Indicating system: Silver wire and mercurous sulfate electrode
Procedure
The silver electrode should be cleaned before beginning the experiment. Rub with a fine grade emery paper and rinse well with distilled water.
Pipet different volumes of each of the halide solutions
into a vessel (total volume ~3 ml), add a few drops of
Obtain an unknown sample and carry out a titration as above.
Treatment of data
For each titration plot E vs V (potential vs volume of titrant) and DE/DV vs V.
Report the composition of the unknown in mM.
Calculates the Ksp of AgCl and AgI (for a point-by-point titration only; explain the restriction). The experimental data are sufficient for this purpose and it is not necessary to know the potential of the reference electrode.
What is the purpose of the Ba(NO3)2?
Recommended
Literature
1. D. A. Skoog, Principles of Instrumental Analysis.
2. D. A. Skoog and D. M. West, Principles of Instrumental Analysis.
3. D.
A. Skoog and J. J. Leary, Instrumental Analysis.
4. D. C. Harris, Quantitative Chemical
Analysis, pp. 250-251, 279-288, 297-300, 351-355, 363-
5. G. D. Christian, Analytical Chemistry.
Go
to Main Page