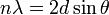X-ray diffraction
The term x-ray diffraction refers both to the physical phenomenon of x-ray scattering in ordered materials as well as a family of analytical techniques which employ such scattering to elucidate structural information about thin films, powders and crystalline material. X-ray diffraction techniques find widespread use in multiple disciplines including biology, chemistry, physics, geology, and materials science. Specialized forms of x-ray diffraction include x-ray crystallography (or single-crystal x-ray diffraction) and powder diffraction. In many cases, corresponding techniques employing neutron or electron diffraction may be used to elucidate similar structural information.
[edit] History
The foundations of experimental x-ray diffraction techniques were laid by Max von Laue in 1913. Von Laue observed that substances which appeared crystalline macroscopically, gave distinctive patterns of reflections when illuminated by a monochromatic x-ray source, including sharp peaks with a high scattering intensity. From the observations of von Laue and their own experiments, father and son Sir William Henry and William Lawrence Bragg hypothesized that the atomic structure of crystalline materials may be considered as repeating layers of regularly spaced ionic planes. Bragg predicted that the high intensity peaks would arise under conditions of specular x-ray reflection where the scattering from multiple planes would lead to constructive interference. The angular position of the peaks, now termed "diffraction peaks" or "Bragg peaks", may be expressed by Bragg's Law:
where λ is the wavelength of the incident radiation, d is the spacing between atomic planes, θ is the incident angle of the incident radiation, and n reflects the order of the reflection. The Braggs were recognized with the Nobel prize in 1915 for their work, which form the fundamental basis for x-ray crystallography and diffraction techniques.[1]
[edit] Structural biology
X-ray diffraction was initially used mostly to solve the structures of small crystalline substances, such as vitamin C by Linus Pauling. The elucidation of the helical DNA structure by James Watson and Francis Crick, based on x-ray data collected by Rosalind Franklin, was a seminal discovery in biology which lead to a Nobel prize. Today, x-ray crystallography is used to elucidate the structure of protein complexes and to elucidate their interaction with each other, their substrates and cofactors. This can help understand the important amino acid residues for function as well as helping determine the different conformations for an enzyme. Most of the structures solved by x-ray crystallography can be obtained freely from the Protein Data Bank.
[edit] References
- ↑ Ashcroft, Neil W., and Mermin, N. David Solid State Physics Orlando, FL: Saunders College Publishing, 1976
| |
Some content on this page may previously have appeared on Citizendium. |