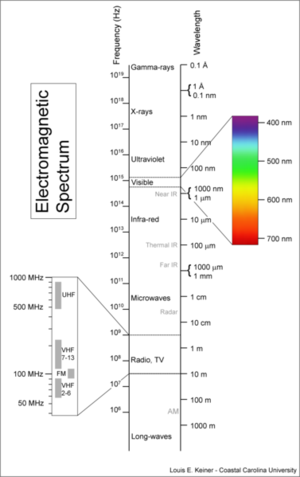
Electromagnetic spectrum
The electromagnetic spectrum is the name given to the range of electromagnetic waves covering all frequencies and wavelengths. It includes radio and television transmission, Microwaves, Infrared, visible light, Ultraviolet, X-rays, and Gamma rays.
Contents |
[edit] Electromagnetic radiation
All electromagnetic radiation can be described in terms of its energy (E), frequency (f), or wavelength (λ). These properties are related by the following equations:
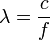 ,
,
 ,
,
where c = 299 792 458 m/s (the speed of light) and h = 6.626 x 10-34 Js (Planck's constant)
Electromagnetic radiation is generally considered to consist of waves. However, in the description of several effects, especially those related to the emission, absorption, and scattering of light, EM radiation seems to consist of particles rather than of waves. That is, the energy carried by EM waves is packaged in discrete bundles called photons or light quanta. The particle character of EM radiation, such as light, is described in quantum electrodynamics, a theory which began with Dirac's work of 1927.[1]
[edit] Electromagnetic spectrum as a resource
Especially in radio and radar frequencies, the electromagnetic spectrum can be a scarce and valuable resource, since a given frequency in a given geographic area can often be used only for one purpose. This is a classic requisite of a technical monopoly. If a frequency is in use for commercial television, than it cannot be used for cellular telephony. In electronic warfare, there may be difficult tradeoffs between letting an enemy use a radio frequency for his own purposes while one's own side is gaining signals intelligence from it, or jamming to deny its use for command and control.
See electromagnetic spectrum management for the policy, management, and commercialization of parts of the spectrum.
[edit] Reference
- ↑ P.A.M. Dirac, Proc. Royal Society (London), The Quantum Theory of the Emission and Absorption of Radiation, vol. A114, p. 243 (1927)
[edit] Further reading
- Young and Freedman. University Physics. Addison-Wesley Publishing Company. ISBN 0-201-31132-1
- NASA introduction to electromagnetic spectrum
| |
Some content on this page may previously have appeared on Citizendium. |