Boltzmann constant
The Boltzmann constant k (also kB) is the ratio of the molar gas constant R to Avogadro's constant NA. It can be thought of as the gas constant for a single molecule (or even for an arbitrary particle in a colloidal solution) rather than for a mole[1].
The Boltzmann constant is illustrated here in the equation for the translational kinetic energy of a simple particle in thermal equilibrium with its surroundings:[2]
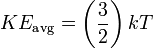
Where KEavg is the average kinetic energy of the particle, k is the Boltzmann constant, and T is the temperature in kelvin.
For more info on this see the equipartition theorem.
According to NIST[3] the Boltzmann constant has a value of 1.3806504 x 10-23 J/K with a standard uncertainty of 0.0000024 x 10-23 J/K and a relative uncertainty of 1.7 x 10-6 (this is represented by the concise form 1.380 6504(24) x 10-23 J/K
The Boltzmann constant can also be represented in alternative units as 8.617385 x 10-5 eV/K
[edit] References
- ↑ Fundamentals of Physics, Fourth Edition by David Halliday, Robert Resnick, and Jearl Walker p582
- ↑ http://hyperphysics.phy-astr.gsu.edu/hbase/kinetic/kintem.html
- ↑ http://physics.nist.gov/cgi-bin/cuu/CCValue?k%7CShowFirst=Browse
| |
Some content on this page may previously have appeared on Citizendium. |

