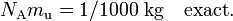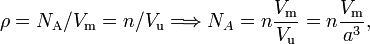Avogadro's number
Avogadro's number, NA, is defined as the number of atoms in 12 gram of carbon-12 atoms in their ground state at rest. By definition it is related to the atomic mass constant mu by the relation
The exact factor 1/1000 appears here by the historic facts that the kilogram is the unit of mass and that in chemistry the mole is preferred over the kmole. The atomic mass constant mu has the mass 1 u exactly (u is the unified atomic mass unit). Avogadro's number is defined here as a number, i.e., a dimensionless quantity. Its latest numeric value[1] is NA = 6.022 141 79 x 1023.
The SI definition of Avogadro's constant (also designated NA) is: the number of entities (such as atoms, ions, or molecules) per mole. (This definition requires a definition of mole that does not rely on NA, but one that is in terms of 12C atoms). In this definition NA has dimension mol−1. The numeric value of Avogadro's constant is NA = 6.022 141 79 x 1023 mol−1.
Because the mole and Avogadro's number are defined in terms of the atomic mass constant (one twelfth of the mass of a 12C atom), Avogadro's constant and number have by definition the same numerical value. In practice the two terms are used interchangeably.
The symbol L is also a recognized SI symbol with the same value as NA.
Avogadro's constant NA is named after the Italian physicist Amedeo Avogadro, and the Loschmidt constant L is named after the Czech-Austrian physicist Josef Loschmidt.
- Remark: It has been argued that the genitive in "Avogadro's constant" is incorrect, because its value was first estimated 10 years after Avogadro's death and the name was given half a century after Avogadro's death. If preferred, one may read "Avogadro constant" and "Avogadro number" everywhere, instead of the corresponding genitive forms used in this article.
Contents |
[edit] History of Avogadro's number
Since 1811, when Amedeo Avogadro put forward his law stating that equal volumes of gas (strictly ideal gas) contain equal number of particles,[2] increasingly sophisticated methods of determining Avogadro’s constant have been developed. These include the kinetic theory of gases, properties of liquid solutions, measurement of the electron charge, black-body radiation, alpha particle emission, and X-ray measurements of crystals.
Without the belief that a macroscopic substance consists of minute particles (initially called atoms, later also molecules), it does not make sense to speak of Avogadro's number. This belief—called atomism— was born in antiquity and grew further in importance with the developments of chemistry early in the 19th century. An important milestone was John Dalton’s law of multiple proportions published in 1804 that gave rise to the first table of the relative weights of atoms. In 1808 Joseph-Louis Gay-Lussac published his law for the combining volumes of gases, namely that gases combine among themselves in very simple proportions of their volumes, and if the products are gases, their volumes are also in simple proportions. For instance, 1 liter of oxygen gas combines with 2 liters of hydrogen gas to form 2 liter of gaseous water. Especially Gay-Lussac's law was of great influence on Avogadro's historical publication of 1811 in which he enunciated his law.
In his 1811 paper Avogadro discusses Gay-Lussac's law and Dalton’s atomic theory. He calculates from gas densities that the molecular weight of nitrogen is 13.238 times the molecular weight of hydrogen (the modern value is 14). Avogadro was the first to propose that the gaseous elements, hydrogen, oxygen, and nitrogen, are diatomic molecules. He deduces that a molecule of water contains a molecule of oxygen and two molecules of hydrogen. Dalton, who had assumed earlier that water is formed from a molecule each of oxygen and hydrogen, rejects Avogadro's and Gay-Lussac's laws.
There are no testimonials that Avogadro ever speculated on the number of molecules in a given gas volume and his law went for a long time largely unnoticed, not in the least because it was not recognized that the law holds strictly only for ideal gases, which many dissociating and associating organic compounds are not. Four years after Avogadro's death, at the historic (1860) chemistry conference in Karlsruhe, his fellow-countryman Stanislao Cannizaro explained why the exceptions to Avogadro's law happen, why the law is important, and why Avogadro deserves the credit for it.
In the beginning of the twentieth century some scientists (the most notable ones being Friedrich Wilhelm Ostwald and Ernst Mach) still denied the existence of molecules. As discussed by Pais,[3] the large number of measurements, based on completely different phenomena, that all led to basically the same value of Avogadro's constant, finally convinced Ostwald of the reality of molecules. Mach died in 1916 as disbeliever. The different experiments for determining NA will be briefly reviewed.
[edit] Estimates from kinetic gas theory
The first estimate of Avogadro’s constant was performed by Loschmidt (1865, 1866).[4] He gave the value 0.969 nm for the diameter of "air molecules" and an equation that relates this diameter to the number L of molecules in one cm3 at standard temperature and pressure. The number L is called Loschmidt’s number; the Avogadro equivalent of Loschmidt's estimate is: NA = 0.4×1023. (Note that at present the SI definitions of L and NA are the same, i.e., the different symbols stand for the same physical quantity). Loschmidt obtained this number by applying the kinetic gas theory of James Clerk Maxwell and Rudolf Clausius, together with experimental data on mean lengths of free paths of molecules in gases and molecular volumes of nitrogen-oxygen compounds. From the latter he estimated the size of the air molecules oxygen and nitrogen. His value 0.969 nm was a factor three too high, and since his equation for L has an inverse-square dependence on the diameter, his estimate for L was an order of magnitude too low. But in any case, Loschmidt was the first to show that Avogadro’s constant is very large and molecules are very small. In 1873 Maxwell used his own kinetic theory of the diffusion coefficient of a gas to obtain a ten times larger value: NA = 4.2×1023.
A simple method for getting the actual volume of molecules is by using the Van der Waals equation (1873) that contains a parameter b, the volume of a single molecule. From b and the volume of the total gas, an estimate of the number of molecules in the gas can be obtained. In the early twentieth century Perrin measured b for mercury vapor, and combining this with results from viscosity measurements, he calculated Avogadro's number to be 6.2×1023,[5] which is a very good value.
[edit] Estimates from liquid solutions
Einstein wrote in his 1905 Ph.D. thesis about the size of molecules and the closely related problem of the magnitude of NA. He derived equations for diffusion coefficients and viscosities in which Avogadro's number appears. From experimental values of the diffusion coefficients and viscosities of sugar solutions in water Einstein gave the estimate NA = 2.1×1023. In a later paper derived from his doctorate work[6] he gave a better estimate from improved experimental data: NA = 4.15×1023, close to Maxwell's value of 1873. Later (1911) it was discovered that Einstein made an algebraic error in his thesis[7] and in the paper based on it. When this was corrected the very same experimental data gave NA = 6.6×1023.
The phenomenon of Brownian motion was first described by Robert Brown in 1828 as the "tremulous motion" of pollen grains (small solid particles of diameter on the order of a micrometer) suspended in water. Einstein's famous 1905 paper on the theory of Brownian motion[8] gives a method for determining NA, but not yet a value.
The first to give a value to NA from Brownian motion was Perrin in 1908. He considered the distribution of Brownian particles in a vertical column in the Earths's gravitational field, and he used a similar mathematical approach to that which leads to the distribution of gas molecules in a vertical column of the atmosphere, see the article on the barometric formula. This formula contains the mass of the particle, the gravitational acceleration g, and the Boltzmann constant k. For suspended Brownian particles one has to make a correction for the buoyancy of the particles in the liquid (Archimedes principle) by using expressions that contain the densities of the particles and the liquid. Measurument of the numbers of particles at two different heights allows the determination of Boltzmann’s constant, k, and Avogadro’s constant through k = NA/R. The molar gas constant R was already known in 1908 with high precision. Perrin in his first experiments prepared a monodisperse colloid of a gum called gamboge. The particle masses were determined by direct weighing of a specified number, and their radii (hence their volumes and densities) by using the Stokes-Einstein law for diffusion. Perrin’s first value for Avogadro’s number was NA = 7.05×1023. In 1909 Perrin [9] coined the name Avogadro's constant when he wrote: Ce nombre invariable N est une constante universelle qu'il semble juste d'appeler constante d'Avogadro [This invariant number N is a universal constant, which may, with justification, be called constant of Avogadro].
[edit] Estimates from the electron charge
Robert Andrews Millikan[10] and his student Harvey Fletcher[11] gave in 1910 and 1911 the first reasonably accurate values for the charge e of the electron. In 1917 Millikan[12] gave the improved value e = 1.591×10−19 C. The current accepted value is the value of the elementary charge 1.6022×10−19 C.
The charge carried by a mole of singly charged ions in an electrochemical cell, which is known as Faraday's constant, F, was already known for quite some time when the electron charge was determined. It was 9.6489×104 C/mol. As F = e ⋅NA, the 1917 value of the electron charge gave Avogadro’s constant as NA = 6.064×1023.
[edit] Estimates from black-body radiation
In 1900 Planck gave birth to quantum theory by showing that the distribution of black body radiation as a function of temperature could be explained by assuming that oscillators in the body of frequency n could only take up or release energy in integer packets of hn, where the proportionality constant h is now known as Planck's constant.
Planck pointed out that a comparison of his theoretical distribution with the experimental curve allowed the determination of h and Boltzmann's constant k. From the ratio of k and the molar gas constant R Avogadro’s constant could be determined. Planck's estimate was NA= 6.175×1023.
[edit] Estimates from counting alpha particles
In 1908, Ernest Rutherford and Hans Geiger[13] concluded that their scintillation technique for detecting α particles (He nuclei) recorded 100% of the particles which are emitted during the radioactive decay of radium. They found that a gram of radium emitted 6.2×1010 particles per second.Counting atoms clearly provides a method for determining Avogadro’s constant. Counting gives the number of a particles produced per second and one only has to measure the volume of helium gas produced per unit of time to know the number atoms per volume, i.e., Avogadro's constant.
In 1911 Rutherford and his friend Boltwood, who spent a year's leave at Rutherford's laboratory in Manchester, measured the amount of helium produced by two radium samples after 83 days and after 132 days, respectively.[14] The first experiment gave 6.58 mm3 of helium gas at 0°C and 760 mm pressure, while the second gave gave 10.38 mm3 of gas. From this the helium production was found to be 2.09x10−2 mm3/day, and 2.03×10−2 mm3/day, which are satisfactorily consistent results. Boltwood and Rutherford did not state the value of Avogadro’s constant, which can be deduced from their experiments and the rate of production of a particles. But, knowing the amount of radium in the sample, and the amount of helium emitted per gram of radium, one can easily deduce that NA = 6.1×1023.
[edit] Estimates from crystal lattice spacings
Although X-rays have been used since 1912 to determine the lattice spacing of crystals, it was not until 1930 that X-ray diffraction was used to determine Avogadro’s constant. Before the 1930s, X-ray wavelengths were not known with enough accuracy, but today lattice spacings of certain crystals, especially the silicon crystal, form the most reliable source of Avogadro's constant.
The number density ρ of a crystal is defined as NA/Vm, where Vm is the molar volume (the volume of one mole). It is reasonable to assume that the number density of the crystal is the same as the number density n/Vu of the unit cell. The volume Vu of the unit cell can be obtained from the lattice spacing of the crystal, provided the geometry of the unit cell is known. The silicon crystal is cubic and has n = 8 atoms per face-centered unit cell of edge length a = 543 pm. Avogadro's number NA follows from
where it is used that Vu = a3 in the case of a cubic unit cell. This equation is the same as equation (143) of Ref.[15]
Since the early 1990s an extensive international effort has been under way to reduce the relative standard uncertainty of the Avogadro constant measured by X-ray diffraction, so that serious consideration can be given to replacing the current SI unit of mass—the international prototype of the kilogram—by a definition based on a natural constant, such as the lattice spacing a of a silicon crystal.
In an outline of the problems, we notice that the exact molar volume Vm of silicon must be known to obtain an exact mole or an exact fraction of a mole. This requires knowledge of the molar mass and hence of the isotopic composition and the amount of impurities of the silicon sample. The three naturally occurring isotopes of Si are 28Si, 29Si, and 30Si, and the amount-of-substance percentages of natural silicon are approximately 92%, 5%, and 3%, respectively. The first problem—the determination of the molar mass—is at present the limiting factor in the accuracy of Avogadro's constant. The second problem is the determination of the molar volume, or, equivalently, of the crystal density ρ. Determination of the lattice spacing a is a third source of error, and, to that end, the wavelength of the X-rays has to be known with great accuracy; this can be obtained from combined optical and X-ray interferometry.
[edit] External links
- An important part of the present article is based on the extensive article by: J. Murrell, Avogadro and his constant
- English translation of Avogadro's 1811 article Essay on a manner ...
- Alfred Bader and Leonard Parker, Joseph Loschmidt, Physicist and Chemist, Physics Today, vol. 54(3), p. 45 (2001) Online
- An Exact Value for Avogadro's Number, American Scientist, March-April 2007 Vol. 95,Online
- Speech by Jean Baptiste Perrin delivered at acceptance of the 1926 Nobel Prize of Physics. Perrin (who was the first to attribute the number NA to Amedeo Avogadro) speaks of "Avogadro's number", not of "the Avogadro constant". Online
[edit] References
- ↑ CODATA value retrieved December 4, 2007 from: Fundamental constants stored at NIST
- ↑ A. Avogadro, Essai d'une manière de déterminer les masses relatives des molécules élémentaires des corps, et les proportions selon lesquelles elles entrent dans ces combinaisons [Essay on a manner of determining the relative masses of the elementary molecules of bodies, and the proportions according to which they enter into these compounds], Journal de Physique, de Chimie et d'Histoire naturelle, vol. 73, pp. 58-76 (1811).
- ↑ A. Pais, Subtle is the Lord ..., Oxford University Press (1982), chapter 5
- ↑ J. Loschmidt, Zur Grösse der Luftmolecüle, Sitzungsberichte der kaiserlichen Akademie der Wissenschaften zu Wien: Math-Naturwiss., Klasse 2, vol. 52, pp. 395–413 (1866). Pre-publication abstract of the author, Anzeiger Kais. Akad. Wiss. Wien, Math-Naturwiss. Klasse 2, 162 (1865).
- ↑ J. B. Perrin, Les Atomes, Paris, Alcan, (1913). Reprint by Gallimard, 1970. Translation: Atoms, by D. L. Hammick, Van Nostrand, New York (1916).
- ↑ A. Einstein, Eine neue Bestimmung der Moleküldimensionen [A new determination of molecule dimensions], Ann. d. Physik, 19, 289, (1906)
- ↑ A. Einstein, Erratum to the 1906 paper, Ann. d. Physik 34, pp. 591–592 (1911)
- ↑ A. Einstein, Über die von der molekularkinetischen Theorie der Wärme geforderte Bewegung von in ruhenden Flüssigkeiten suspendierten Teilchen [On the motion, required by the molecular kinetic theory of heat, of particles suspended in liquids at rest], Ann. d. Physik, 17, 549, (1905)
- ↑ Jean Perrin, Mouvement brownien et réalité moléculaire, Ann. Chim. Phys. vol. 18, pp. 1–114 (1909).Online
- ↑ R.A. Millikan, A new modification of the cloud method of determining the elementary electrical charge and the most probable value of that charge, Phil. Mag., vol. 19, pp. 209-228 (1910)
- ↑ H.Fletcher, A Verification of the Theory of Brownian Movements and a Direct Determination of the Value of Ne For Gaseous Ionization, Phys. Rev., vol. 33, pp. 81-110 (1911)
- ↑ R. A. Millikan, The Electron: Its Isolation and Measurements and the Determination of Some of Its Properties, University of Chicago Press, Chicago (1917).
- ↑ E. Rutherford and H. Geiger, An Electrical Method of Counting the Number of α-Particles from Radio-Active Substances, Proc. Roy. Soc., vol. A81, pp. 141-161. (1908)
- ↑ B.B. Boltwood and E. Rutherford, Production of helium by radium, Phil.Mag., vol. 22, pp. 586-604 (1911)
- ↑ P.J. Mohr and B. N. Taylor, CODATA recommended values of the fundamental physical constants: 2002, Reviews Modern Physics, vol. 77, p. 1 (2005)
Further reference:
- J. Wisniak, Amedeo Avogadro The Man, the Hypothesis, and the Number, Chem. Educator, vol. 5, pp. 263-268 (2000) (This paper errs in giving the value for Avogadro's constant obtained by Loschmidt. It gives it a factor ten too high).