Answer to the Question 12/96
The question was:When a coffee drop dries out, the coffee doesn't stay uniform; instead it concentrates in a ring around the boundary. The same thing happens when almost any drop dries. What makes the coffee become so nonuniform?
No complete unswer was submitted. Here is a brief explanation, by Rob Deegan (the original appears on the web):
Why do drying drops form a ring?
 The image to the left is a common household sight: a dried coffee stain.
It is immediately appearent from the ring-like appearance of the stain that
the majority of the residue is concentrated along the perimeter. This is
intriguing because it indicates that some physical or chemical process is
at work in drying drops which is able to segregate the solute from the
solvent. At the University of Chicago, we attempted to answer the
questions of how general is this phenomenon, what is driving mechanism
responsible for it, and are there practical technological implications?
The image to the left is a common household sight: a dried coffee stain.
It is immediately appearent from the ring-like appearance of the stain that
the majority of the residue is concentrated along the perimeter. This is
intriguing because it indicates that some physical or chemical process is
at work in drying drops which is able to segregate the solute from the
solvent. At the University of Chicago, we attempted to answer the
questions of how general is this phenomenon, what is driving mechanism
responsible for it, and are there practical technological implications?
We have determined that ring formation is a ubiquitous and robust phenomenon. It doesn't depend on the solute, the solvent, or the substrate so long as the solvent is partially wetting and volatile, and the contact line is pinned.
Traditional mechanism of solute transport--Marangoni flow, Rayleigh-Benard convection, diffusion, electrostatic and electrokinetic effects, and wetting phenomenon--do not account for ring formation. The phenomenon is due to a previously unexplored form of capillary flow: the contact line of the drying drop is pinned such that liquid evaporating from the edge must be replenished by liquid from the interior.
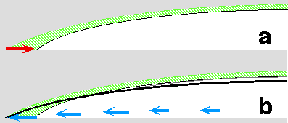 This mechanism is illustrated in the schematic to the right. Pictures a and b show a cross section of a drop during an infinitesimal increment of evaporation. The green hatched area represents the volume of liquid removed by evaporation in a single time step. The liquid removed from the edge must be replenished by an outward flow (indicated in blue).
This mechanism is illustrated in the schematic to the right. Pictures a and b show a cross section of a drop during an infinitesimal increment of evaporation. The green hatched area represents the volume of liquid removed by evaporation in a single time step. The liquid removed from the edge must be replenished by an outward flow (indicated in blue).
We are also able to qualitatively alter the morphology of the ring deposit. Since evaporation is the driving mechanism, we can produce different deposits by altering the evaporation profile.
 | In the figure to the left, we indicate schematically three evaporation profiles by the amount of liquid removed at a given radius (red hatched lines) and a corresponding photograph of the resulting deposit. Interestingly, when the evaporation profile is such that no water leaves from the edge (bottom profile), no ring is formed. This qualitatively demonstrates the validity of the above mechanism. |
|
As an experimental system, we used a colloidal suspension of polystyrene spheres in water. Fluorescent video microscopy was used to image the spheres, and image analysis techniques were used to extract information about the concentration and flow of the spheres in the bulk and their accumulation in the ring. We are able, using the simple mathematical model of the mechanism introduced above, to accurately predict these quantities (see pre-print).
|  |
 Ring deposition is a potentially useful
technology impacting printing, washing and coating processes. We are pursuing
two application possibilities: fine wire assembly and DNA stretching. Using a
colloidal suspension of gold particles it might be possible to assemble a fine
wire with dimensions comparable to those achieved with modern lithographic
methods. It might also be possible to stretch DNA using the strong shear flow
that develops in ring forming drops when the evaporation rate is enhanced at the
edge.
Ring deposition is a potentially useful
technology impacting printing, washing and coating processes. We are pursuing
two application possibilities: fine wire assembly and DNA stretching. Using a
colloidal suspension of gold particles it might be possible to assemble a fine
wire with dimensions comparable to those achieved with modern lithographic
methods. It might also be possible to stretch DNA using the strong shear flow
that develops in ring forming drops when the evaporation rate is enhanced at the
edge.
"Capillary flow as the cause of ring stains from dried liquid drops"
Robert D. Deegan, Olgica Bakajin, Todd F. Dupont, Greg Huber, Sidney R. Nagel, and Thomas A. Witten, was published in Nature.
 Back to "front page"
Back to "front page"